Drug Enforcement Administration
From Rx-wiki
The Drug Enforcement Administration (DEA) is a United States Department of Justice law enforcement agency, a federal police service tasked with enforcing the Controlled Substances Act of 1970. Not only is the DEA the lead agency for domestic enforcement of federal drug laws (sharing concurrent jurisdiction with the Federal Bureau of Investigation), it also has sole responsibility for coordinating and pursuing U.S. drug investigations abroad.
The DEA was created by President Richard Nixon through an Executive Order in July 1973 in order to establish a single unified command to combat "an all-out global war on the drug menace." At its outset, the DEA had 1,470 Special Agents and a budget of less than $75 million. Furthermore, in 1974, the DEA had 43 foreign offices in 31 countries. Today, the DEA has approximately 5,000 Special Agents, a budget of more than $2 billion and 86 foreign offices in 62 countries.
Contents
Mission Statement
The mission of the DEA is to enforce the controlled substances laws and regulations of the United States and bring to the criminal and civil justice system of the United States, or any other competent jurisdiction, those organizations and principal members of organizations, involved in the growing, manufacture, or distribution of controlled substances appearing in or destined for illicit traffic in the United States; and to recommend and support non-enforcement programs aimed at reducing the availability of illicit controlled substances on the domestic and international markets.
In carrying out its mission as the agency responsible for enforcing the controlled substances laws and regulations of the United States, the DEA's primary responsibilities include:
- Investigation and preparation for the prosecution of major violators of controlled substance laws operating at interstate and international levels.
- Investigation and preparation for prosecution of criminals and drug gangs who perpetrate violence in our communities and terrorize citizens through fear and intimidation.
- Management of a national drug intelligence program in cooperation with federal, state, local, and foreign officials to collect, analyze, and disseminate strategic and operational drug intelligence information.
- Seizure and forfeiture of assets derived from, traceable to, or intended to be used for illicit drug trafficking.
- Enforcement of the provisions of the Controlled Substances Act as they pertain to the manufacture, distribution, and dispensing of legally produced controlled substances.
- Coordination and cooperation with federal, state and local law enforcement officials on mutual drug enforcement efforts and enhancement of such efforts through exploitation of potential interstate and international investigations beyond local or limited federal jurisdictions and resources.
- Coordination and cooperation with federal, state, and local agencies, and with foreign governments, in programs designed to reduce the availability of illicit abuse-type drugs on the United States market through nonenforcement methods such as crop eradication, crop substitution, and training of foreign officials.
- Responsibility, under the policy guidance of the Secretary of State and U.S. Ambassadors, for all programs associated with drug law enforcement counterparts in foreign countries.
- Liaison with the United Nations, Interpol, and other organizations on matters relating to international drug control programs.
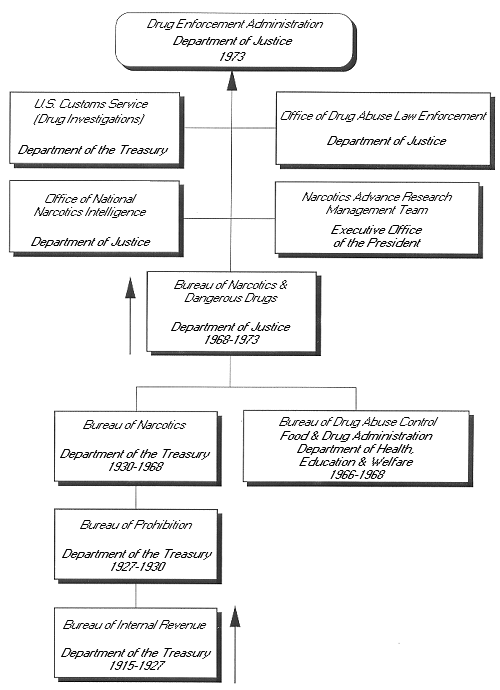
Genealogy
Medication Schedules
Controlled substances are medications with further restrictions due to abuse potential. There are 5 schedules of controlled substances with various prescribing guidelines based on abuse potential counter balanced by potential medicinal benefit as determined by the Drug Enforcement Administration and individual state legislative branches. The DEA is provided with this authority by the Controlled Substances Act. Below is a brief explanation of the schedules along with example medications.
Schedule I (CI)
- Characteristics:
- Unaccepted medical use.
- Highest potential for abuse.
- Not available by a prescription.
- Examples:
- LSD
- heroin
- Quaaludes (methaqualone)
Schedule II (CII)
- Characteristics:
- High potential for abuse or misuse.
- Sufficient medicinal use to justify availability as a prescription.
- Examples:
- oxycodone
- morphine
- amphetamines
Schedule III (CIII)
- Characteristics:
- Potential risk for abuse, misuse, and dependence.
- Examples:
- Vicodin (hydrocodone bitartrate and acetaminophen)
- Codeine and codeine containing products in a solid dosage form (tablet, capsule, etc.).
Schedule IV (CIV)
- Characteristics:
- Low potential for abuse and limited risk of dependence.
- Examples:
- phenobarbital
- benzodiazepines
- other sedatives and hypnotics
Schedule V (CV)
- Characteristics:
- Low potential for abuse or misuse.
- Examples:
- Cough medicines that contain a limited amount of codeine.
- Antidiarrheal medications that contain a limited amount of an opiate such as Lomotil (diphenoxylate and atropine).
CI medications are not available via a prescription
CII medications may be written for a maximum 90 day supply without refills
CIII – CV medications may be written for up to a 6 month supply
Many problems associated with drug abuse are the result of legitimately-manufactured controlled substances being diverted from their lawful purpose into the illicit drug traffic. Many of the narcotics, depressants and stimulants manufactured for legitimate medical use are subject to abuse, and have therefore been brought under legal control. The goal of controls is to ensure that these "controlled substances" are readily available for medical use, while preventing their distribution for illicit sale and abuse.
Prescription drug abuse facts
- Nearly 7 million Americans were abusing prescription drugs in 2006—more than the number who are abusing cocaine, heroin, hallucinogens, Ecstasy, and inhalants, combined. That 7 million was just 3.8 million in 2000, an 80 percent increase in just 6 years.
- Prescription pain relievers are new drug users’ drug of choice, vs. marijuana or cocaine.
- Opioid painkillers now cause more drug overdose deaths than cocaine and heroin combined.
- 1 in 7 teens admit to abusing prescription drugs to get high in the past year. Sixty percent of teens who abused prescription pain relievers did so before the age of 15.
- 2 in 5 teens believe that prescription drugs are “much safer” than illegal drugs. And 3 in 10 teens believe that prescription pain relievers are not addictive.
- Misuse of painkillers represents three-fourths of the overall problem of prescription drug abuse; hydrocodone is the most commonly diverted and abused controlled pharmaceutical in the U.S.
- Twenty-five percent of drug-related emergency department visits are associated with abuse of prescription drugs.
- Methods of acquiring prescription drugs for abuse include “doctor-shopping,” traditional drug-dealing, theft from pharmacies or homes, illicitly acquiring prescription drugs via the Internet, and from friends or relatives.
- DEA works closely with the medical community to help them recognize drug abuse and signs of diversion and relies on their input and due diligence to combat diversion. Doctor involvement in illegal drug activity is rare—less than one tenth of one percent of more than 750,000 doctors are the subject of DEA investigations each year—but egregious drug violations by practitioners unfortunately do sometimes occur. DEA pursues criminal action against such practitioners.
- DEA Internet drug trafficking initiatives over the past several years have identified and dismantled organizations based both in the U.S. and overseas, and arrested dozens of conspirators. As a result of major investigations such as Operations Web Tryp, PharmNet, Cyber Rx, Cyber Chase, and Click 4 Drugs, Bay Watch, and Lightning Strike, tens of millions of dosage units of prescription drugs and tens of millions of dollars in assets have been seized.
Diversion control system
Many problems associated with drug abuse are the result of legitimately-manufactured controlled substances being diverted from their lawful purpose into the illicit drug traffic. Many of the narcotics, depressants and stimulants manufactured for legitimate medical use are subject to abuse, and have therefore been brought under legal control. The goal of controls is to ensure that these "controlled substances" are readily available for medical use, while preventing their distribution for illicit sale and abuse.
Under federal law, all businesses which manufacture or distribute controlled drugs, all health professionals entitled to dispense, administer or prescribe them, and all pharmacies entitled to fill prescriptions must register with the DEA. Authorized registrants receive a "DEA number". Registrants must comply with a series of regulatory requirements relating to drug security, records accountability, and adherence to standards.
A DEA number is a series of numbers assigned to a health care provider (such as a dentist, physician, nurse practitioner, or physician assistant) allowing them to write prescriptions for controlled substances. Legally the DEA number is solely to be used for tracking controlled substances. The DEA number, however, is often used by the industry as a general "prescriber" number that is a unique identifier for anyone who can prescribe medication.
A valid DEA number consists of:
- 2 letters and 7 digits
- The first letter is always an A (deprecated), B (most common), or F (new) for a dispenser. (Also M is available for mid-level practitioners in some states and either P or R is used for a wholesaler)
- The second letter is typically the initial of the registrant's last name
- The seventh digit is a "checksum" that is calculated as:
- Add together the first, third and fifth digits
- Add together the second, fourth and sixth digits and multiply the sum by 2
- Add the above 2 numbers
- The last digit (the ones value) of this last sum is used as the seventh digit in the DEA number
See also
Federal pharmacy law
Comprehensive Drug Abuse Prevention and Control Act
Combat Methamphetamine Epidemic Act
Food and Drug Administration
References
- DEA, DEA History, http://www.justice.gov/dea/history.htm
- DEA, DEA Staffing and Budget, http://www.justice.gov/dea/agency/staffing.htm
- DEA, DEA Mission Statement, http://www.justice.gov/dea/agency/mission.htm
- DEA, Genealogy, http://www.justice.gov/dea/agency/genealogy.htm
- DEA, Fact Sheet: Prescription Drug Abuse - A DEA Focus, http://www.justice.gov/dea/concern/prescription_drug_fact_sheet.html
- Drug Trends, presented by DEA special agent Mike Kupchik, September 7,2011
- Pharmacy Times, A Review of Federal Legislation Affecting Pharmacy Practice, Virgil Van Dusen , RPh, JD and Alan R. Spies , RPh, MBA, JD, PhD, https://secure.pharmacytimes.com/lessons/200612-01.asp
- Strauss's Federal Drug Laws and Examination Review, Fifth Edition (revised), Steven Strauss, CRC Press, 2000
- Pharmacy Times, An Overview and Update of the Controlled Substances Act of 1970, Virgil Van Dusen , RPh, JD and Alan R. Spies , RPh, MBA, JD, PhD, https://secure.pharmacytimes.com/lessons/200702-01.asp
- Drug Enforcement Administration Office of Diversion Control, Title 21 United States Code (USC) Controlled Substances Act, http://www.deadiversion.usdoj.gov/21cfr/21usc/index.html
- Controlled Substance Ordering System, http://www.deaecom.gov/
- Drug Enforcement Administration Office of Diversion Control, Practitioner's Manual SECTION V – VALID PRESCRIPTION REQUIREMENTS, http://www.deadiversion.usdoj.gov/pubs/manuals/pract/section5.htm