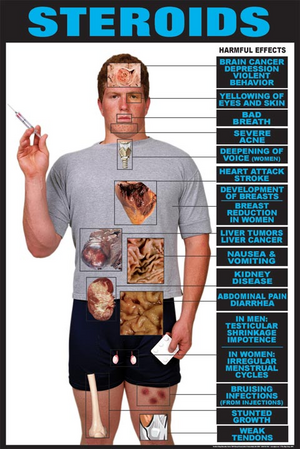
Anabolic Steroids Control Act
From Rx-wiki
The intent of the ASCA is to minimize (or eliminate) the use of anabolic steroids for non-medical purposes. Prior to the ASCA, anabolic steroids were regulated as drugs pursuant to the FDCA.
The DEA has classified anabolic steroids as Schedule III controlled substances; however, certain products which are considered to have no significant potential for abuse because of their concentration, preparation, mixture, or delivery system, are exempt from being classified as control substances (such as Premarin with methyltestosterone tablets).
In addition, the classification does not include anabolic steroids expressly intended for cattle or other non-human species and which are approved by the FDA. However, prescribing, dispensing or distributing such substances for other than implantation in cattle or other non-human species is a violation of the law.
See also
Federal pharmacy law
Drug Enforcement Administration
References
- Pharmacy Times, A Review of Federal Legislation Affecting Pharmacy Practice, Virgil Van Dusen , RPh, JD and Alan R. Spies , RPh, MBA, JD, PhD, https://secure.pharmacytimes.com/lessons/200612-01.asp
- Strauss's Federal Drug Laws and Examination Review, Fifth Edition (revised), Steven Strauss, CRC Press, 2000
- Drug Enforcement Administration Office of Diversion Control, Title 21 United States Code (USC) Controlled Substances Act, http://www.deadiversion.usdoj.gov/21cfr/21usc/index.html
- "Steroids", http://students.cis.uab.edu/archived/zmabeus/long-term.html